Our caring team at the OrthoBiologics comprises of dedicated professionals and physiotherapists who provide you with state-of-the-art treatments for various bone and joint disorders including arthritis, joint pain, and sports injuries. We offer regenerative services which includes platelet-rich plasma (PRP) injections, stem cell therapy, nSTRIDE APS. To know more about our services, read below:
Platelet-Rich-Plasma
PRP is an innovative treatment that uses concentrated growth factors from your own blood to treat soft tissue injuries and arthritis.
Stem Cells
Stem Cells are a powerful and unique treatment for patients with osteoarthritis, cartilage defects and slow-healing fractures.
nSTRIDE APS
nSTRIDE autologous protein solution (APS) is a ground-breaking new treatment for patients with arthritis. The substance is obtained from your blood, specially concentrated, and then injected into the affected joint. Research has shown excellent and ongoing improvements in pain levels for up to 2 years.
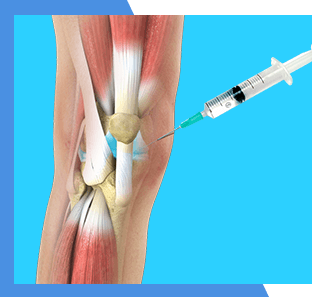
Viscosupplementation
Viscosupplementation uses a concentrated form of a natural joint lubricant to help patients with mild to moderate arthritis.
Combination Therapy for the Treatment of Osteoarthritis
Research has shown the benefits of combining various Orthobiologics treatments with each other. This can enhance the effect of the treatment and improve the long-term outcome of the procedure.
If you wish to be advised on the most appropriate treatment, please call to schedule an appointment or click to request an appointment online.





